SPORTS SCIENCE EXCHANGE
ANTI-INFLAMMATORY DRUGS, KIDNEY FUNCTION, AND EXERCISE
SSE#67- Volume 11 (1997), Number 4
Bill Farquhar, M.S.
Noll Physiological Research Center
The Pennsylvania State University
University Park, PA
W. Larry Kenney, Ph.D.
Noll Physiological Research Center
The Pennsylvania State University
University Park, PA
Member, GSSI Sports Medicine Review Board
KEY POINTS
1. The use of nonsteroidal anti-inflammatory drugs (NSAIDs) before and during exercise has been linked to acute renal failure in a handful of case-study reports. Similarly, long-term NSAID may be involved in some cases of chronic renal failure, independent of exercise.
2. Acute renal failure occurs when the kidney is unable to maintain fluid and electrolyte balance and excrete waste products.
3. NSAIDs act therapeutically by inhibiting prostaglandin synthesis thereby reducing pain and inflammation. Because renal prostaglandins protect against decreases in renal blood flow during exercise, renal PG inhibition with an NSAID during exercise may lead to an abnormally depressed renal blood flow and glomerular filtration rate.
4. Excessive NSAID use may increase the potential for renal problems associated with exercise. This potential is magnified if prolonged exercise is combined with severe heat stress and/or dehydration.
5. Fortunately, the incidence of renal failure associated with exercise is low, even in athletes who habitually use NSAIDs.
6. Proper hydration prior to and throughout exercise can minimize any risk that NSAIDs may pose to the kidney.
INTRODUCTION
Nonsteroidal anti-inflammatory drugs (NSAIDs) are used by competitive athletes and recreational exercisers because of their analgesic (pain reduction) and anti-inflammatory benefits. A list of common NSAIDs and other analgesics available over-the-counter (OTC) is presented in Table 1 (Physicians' Desk Reference, 1997). Estimates indicate that approximately 50 million Americans intermittently or regularly use NSAIDs, making this class of drugs one of the most widely used across all age groups. The most recent annual data available (year ending June 22, 1997) indicate that OTC analgesics accounted for $2.693 billion in sales (Information Resources Inc., Chicago, 1997).
Case study reports have linked NSAIDs to acute renal failure during exercise. For example, Macsearraigh et al. (1979) reported that 7 out of the 9 runners who developed renal failure during a 9-year period were taking some form of analgesic. In addition, all four runners who developed renal failure during the 1986 Comrades Marathon in South Africa were taking NSAIDs (Seedat et al., 1990). The Comrades Marathon is an example of an extreme endurance event, covering 90-kilometers (approximately 56 miles) between the cities of Durban and Pietermaritzburg. The course is hilly, and the race takes place in May when the weather is typically warm. There have been similar reports of runners developing acute renal failure in other races, often under less severe conditions. For example, a 41-year-old male marathon runner developed acute renal failure after competing in a race following naproxen use (Vitting et al., 1986).
The association between NSAID use and renal failure is not limited to exercise conditions. Whelton (1995) recently reviewed individual case reports of NSAID-induced acute renal failure. A common finding is the presence of risk factors such as chronic heart failure, renal disease, and hepatic disease. Outside of these chronic disease states (which do not tend to affect young and healthy athletes), Whelton reported that alcohol consumption (binge drinking) in combination with NSAID use may cause renal failure in younger individuals. Although there are obvious limitations when attempting to draw conclusions based solely on case studies, these reports are alarming and warrant attention.
Epidemiological reports also link long-term NSAID use with end-stage renal disease (ESRD). A retrospective case-control study that surveyed 716 patients with ESRD and 361 apparently healthy control subjects found that heavy NSAID use -more than 5000 pills in a lifetime- was associated with a nine-fold greater rate of ESRD (Perneger et al., 1994). Since it remains to be determined if the mechanisms causing NSAID-induced long-term renal dysfunction and acute renal failure are the same, the remainder of this review will focus only on the literature dealing with acute changes in renal function during exercise and related stresses.

Acute Renal Failure
The renal failure that may occur with NSAID use results from a reduction in renal perfusion, leading to a depressed glomerular filtration rate (GFR). Acute renal failure can be characterized by the inability of the kidney to maintain fluid and electrolyte balance and to eliminate nitrogenous waste products. Urinary output is usually less than 400 mL per day (oliguric renal failure) but can be greater (nonoliguric renal failure; > 400 mL per day). Renal failure can be defined by a number of different criteria, including an increase in serum creatinine of > 0.5 mg/dL (44 mmol/L) over baseline values, a reduction in creatinine clearance of > 50%, or a decrease in renal function that results in the need for dialysis. Prompt dialysis can prevent uremia, hyperkalemia, and other complications of fluid overload (Thadhani et al., 1996).
Renal Function During Exercise
During exercise in the heat, skin and active muscle tissue compete for a limited cardiac output. The increased blood flow to the skin along with the evaporation of sweat allows heat to be dissipated to the environment while increased blood flow to the muscle allows for the delivery of oxygen and energy substrates. In order to accomplish this dual purpose without a decrease in blood pressure, blood flow to the liver, pancreas, gastrointestinal tract, and kidney decreases (Rowell, 1993). When the intensity of exercise exceeds 50% VO2max, blood flow to the kidney (RBF), filtration rate of the kidney (glomerular filtration rate; GFR), sodium excretion and urine flow rate all decrease by 30-60% (Zambraski et al., 1996). Additional perturbations such as heat stress, sodium depletion, and dehydration may exaggerate these decrements in renal function. For example, Smith et al. (1952) found that GFR did not change during very-light-intensity treadmill exercise (3 mph, 5% grade) performed in thermoneutral conditions. When this exercise was performed in the heat (50°C), GFR decreased 19%, and when heat and dehydration (-3.5% to -7.7% loss in body mass) were combined, GFR decreased 51%.
These changes in renal function during exercise are caused mainly by enhanced activity of the sympathetic nervous system that causes renal vasoconstriction (Zambraski, 1996). Increases in antidiuretic hormone (ADH), renin, angiotensin II (ANGII), and aldosterone also contribute (see Figure 1). The end results are increased sodium and water conservation by the kidneys and the maintenance of mean arterial pressure during exercise. While these adjustments are beneficial for homeostasis, excessive reductions in renal function can precipitate renal failure.
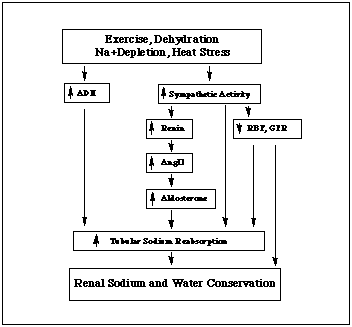 |
| Figure 1. Factors controlling renal function during acute exercise that result in the renal conservation of sodium and water (redrawn from Zambraski, 1996). |
NSAIDs, Prostaglandins, and the Kidney
NSAIDs act therapeutically by inhibiting the enzyme cyclooxygenase, thereby preventing the breakdown of arachidonic acid to prostaglandins (PGs). Some of the PGs -- specifically PGE2 and PGI2 -- cause vasodilation in the kidney (Murray & Brater, 1993) and may also be involved in the release of renin (Kramer et al., 1985). In young, healthy subjects during unstressed conditions, basal renal vasoconstrictor tone is low, and the vasodilation produced by PGs is not required for normal kidney function (Sedor et al., 1986). Therefore, when OTC or prescription doses of NSAIDs are ingested, there are no apparent adverse renal effects. This is not the case during disease states such as heart failure (Dzau et al., 1984) or liver cirrhosis (Zipser et al., 1979). Renal vasoconstriction in these disease states results from increased sympathetic outflow to the kidney, increased blood catecholamine concentrations, and the production of renin-angiotensin II. These are the same physiological changes that occur during exercise. The PGs may therefore be predicted to become more important mediators of renal function during exercise because of this heightened vasoconstrictor tone and the consequent reductions in RBF and GFR. This mechanism of NSAID inhibition (illustrated in Figure 2) of PG function during exercise may explain the case-study reports that link NSAID use and acute renal failure.
|
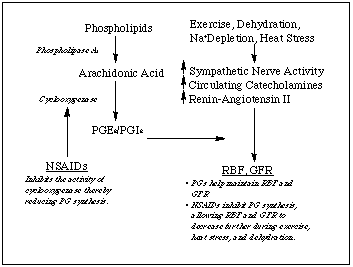
|
| Figure 2. The mechanisms by which NSAIDs affect renal function. |
There are only a few laboratory studies that have examined this relationship. Zambraski et al. (1982) had six healthy men exercise for 30 min at 70% VO2max in a hydrated (4 mL/kg of water before the trial) and a hypohydrated state (no food and water for 10 h before the trial). The exercise trials were conducted under thermoneutral conditions, and each subject completed two trials, one trial after taking aspirin (3.25 g/d for 3 d prior to testing) and one control trial (no aspirin). No differences were found in creatinine clearance (used as an index of GFR) when the aspirin trials were compared to the control trials in either hydration state. This led the authors to conclude that aspirin treatment had no significant effects on the renal response to short-term exercise. In another study (Walker et al., 1994), eight men exercised for 30 min at a slightly greater exercise intensity (80% VO2max) with and without prior indomethacin intake (50 mg every 8 h for 36 h prior to the exercise). All subjects were well hydrated (20 mL/kg) prior to exercise. Even at this slightly greater exercise intensity, there were again no significant differences in GFR (by inulin clearance), but RBF was significantly lower in the indomethacin trial compared to the control trial. Because of the lower RBF, the authors concluded that NSAIDs such as indomethacin may impair renal function during exercise conditions. Because the main function of the kidney is to filter the blood and because both studies reported no significant changes in GFR during the NSAID trials, the question of whether NSAID use inhibits GFR under more severe exercise conditions, which many athletes typically encounter, remained unaddressed.
We recently completed a study that simulated the conditions that an endurance athlete might expect to encounter during the summer months (Farquhar et al., 1997). This entailed reducing bodily stores of sodium and water in 12 healthy men and women prior to a 45-min run at 65% VO2max in a temperature controlled (36°C) environmental chamber. Each subject completed three trials: a placebo trial, a trial in which the maximal OTC dose of acetaminophen was given, and a trial in which the maximal OTC dose of ibuprofen was given. [Ibuprofen and acetaminophen were employed because they are both commonly used analgesics and antipyretics. Ibuprofen also has anti-inflammatory actions at higher prescription doses.] The sodium- and volume-depletion were accomplished with a salt-restricted diet for 3 d and a 60-min dehydration period before exercise. We hypothesized that this sequential increase in renal stressors (salt restriction, dehydration, and exercise in the heat) would render the kidneys PG-dependent and that ibuprofen would cause greater transient reductions in GFR (compared to the placebo trial). Acetaminophen is thought to be a very weak inhibitor of PG synthesis in peripheral tissues; therefore, it was hypothesized that acetaminophen would not cause further reductions in renal function. However, recent data have challenged the premise that acetaminophen has minimal effects in peripheral tissues such as the kidney. Using a renal PG-dependent animal model, Zambraski et al. (1996) demonstrated that acetaminophen inhibited production of renal PGs and caused decrements in RBF and GFR, although not to the same extent as ibuprofen.
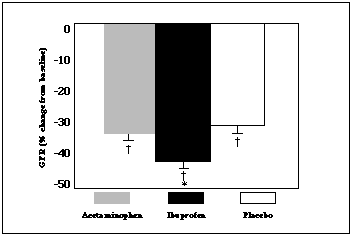 |
| Figure 3. The % change in GFR from initial baseline values immediately following an exercise bout in the heat. Subjects were dehydrated before the start of exercise. The GFR was significantly decreased in all three trials (Ý = p |
The data confirmed our initial hypothesis, that is, OTC ibuprofen use was associated with significantly greater reductions in GFR during exercise in this sodium and volume-depleted condition (Figure 3). The reductions in GFR during the acetaminophen trials were not different from the placebo trials. The ibuprofen-induced reductions in GFR were modest, but under conditions where the exercise intensity might be greater and/or the degree of dehydration might be more severe, ibuprofen use could potentially precipitate acute renal deficiencies or possibly failure.
Significance
Despite the previously discussed case study reports and the suggestion that NSAID use inhibits GFR, the incidence of acute renal failure during exercise is remarkably low. This fact is made even more impressive when one considers the large number of athletes taking NSAIDs prior to and during endurance events. It is important to keep in mind that even though many NSAIDs inhibit renal PGs, most are only able to inhibit PG production by approximately 50-60% (Zambraski & Dunn, 1993). This means that some PG production continues, helping assure the maintenance of renal hemodynamics. Also, other substances such as nitric oxide may also preserve renal vasodilation and help prevent excessive decreases in GFR (Deng et al., 1996), although the exact role of nitric oxide in the kidney remains to be elucidated. These factors make it difficult to unequivocally quantify a precise role for renal PGs during exercise.
Practical Recommendations
Even though the incidence of renal failure during exercise is low, it would seem prudent for athletes to use NSAIDs judiciously during training and competition. Since the scientific literature suggests that athletes who exercise in hot environments in a hypohydrated state are likely to have greater reductions in GFR, adequate hydration prior to and during exercise would minimize any risk. Also, although it has not been studied, very high doses of a NSAID could theoretically have even greater effects on renal function and thereby expose the athlete to greater risk. Therefore, athletes should not exceed the OTC dose unless directed to do so by a physician. Physicians should stress the importance of proper hydration during exercise, especially when higher, prescription doses of NSAIDs are utilized for prevention or treatment of inflammation. Where appropriate, non-NSAID analgesics such as acetaminophen should be considered.
Summary and Conclusions
Use of NSAIDs during prolonged exercise has been linked to acute renal failure in case-study reports. These NSAIDs may depress renal function by inhibiting the cyclooxygenase enzyme, causing a decrease in the production of PGs. The PGs are thought to aid in the maintenance of renal hemodynamics during exercise, i.e., to modulate precipitous falls in GFR and RBF. Although the overall incidence of acute renal failure among athletes is low, caution should be exercised when NSAIDs are used in concert with dehydrating exercise in the heat. Proper hydration before and during exercise may help to prevent severe reductions in renal function and minimize the risk of renal failure.
References
Deng, X., W.J. Welch, & C.S. Wilcox (1996). Role of nitric oxide in short-term and prolonged effects of angiotensin II on renal hemodynamics. Hypertension 27:1173-1179.
Dzau, V.J., M. Packer., L.S. Lilly., S.L. Swartz., N.K. Hollenberg, & G.H. Williams (1984). Prostaglandins in severe congestive heart failure-relation to activation of the renin-angiotensin system and hyponatremia. N Engl J Med 310:347-352.
Farquhar, W.B., A.L. Morgan, E.J. Zambraski, & W.L. Kenney (1997). Effects of acetaminophen vs. ibuprofen on renal function and hemodynamics in the stressed kidney (abstract). FASEB J. 11:A87.
Information Resources, Inc. Chicago, 1997.
Kramer, H.J., B. Stinnesbeck., G. Klautke., J. Kipnowski., D. Klingmueller., K. Glaenzer, & R. Duesing (1985). Interaction of renal prostaglandins with the renin-angiotensin and renal adrenergic nervous system in healthy subjects during dietary changes in sodium intake. Clin. Sci. 68:387-393.
MacSearraigh, E.T.M., J.C. Kallmeyer, & H.B. Schiff (1979). Acute renal failure in marathon runners. Nephron 24:236-240.
Murray, M.D., & D.C. Brater (1993). Renal toxicity of the nonsteroidal anti-inflammatory drugs. Annu. Rev. Pharmacol. Toxicol.32:435-465.
Perneger, T.V., P.K. Whelton, & M.J. Klag (1994). Risk of kidney failure associated with the use of acetaminophen, aspirin, and nonsteroidal anti-inflammatory drugs. N Engl J Med 331:1675-1679.
Physicians' Desk Reference for Non-prescription Drugs (18th ed.). Montvale, NJ: Medical Economics Company, Inc., 1997.
National Disease and Therapeutic Index. Antiarthritic class (09100). Ambler, Pennsylvania: IMS America; 1987.
Rowell, L.B. (1993). Human Cardiovascular Control. New York: Oxford University Press.
Sedor, J.R., E.W. Davidson, & M.J. Dunn (1986). Effects of nonsteroidal anti-inflammatory drugs in healthy subjects. The Am. J. Med. 81(Suppl 2B):58-70.
Seedat, Y.K., N. Aboo., S. Naicker, & I. Parsoo (1990). Acute renal failure in the "Comrades Marathon" runners. Renal Failure 11:209-212.
Smith, J.H., S. Robinson, & M. Pearcy (1952). Renal responses to exercise, heat, and dehydration. J. Appl. Physiol. 4:659-665.
Thadhani, R., M. Pascual, & J.V. Bonventre (1996). Acute renal failure. N Engl J Med 334:1448-1460.
Vitting, K.E., N.J. Nichols, & G.R. Seligson (1986). Letter to the editor: Naproxen and acute renal failure in a runner. Ann. Intern. Med.105:144.
Walker, R.J., J.P. Fawcett, E.M. Flannery, & D.F. Gerrard (1994). Indomethacin potentiates exercise-induced reduction in renal hemodynamics in athletes. Med. Sci. Sports Exerc. 26:1302-1306.
Whelton, A. (1995). Renal effects of over-the-counter analgesics. J. Clin. Pharmacol. 35:454-463.
Zambraski, E.J. (1996). Body Fluid Balance. Boca Raton, FL: CRC Press, Inc.
Zambraski, E.J., A. Colletti, T. Rahe, & H. Vogl (1996). Effects of acetaminophen versus ibuprofen on renal function in normal and sodium depleted subjects (abstract). Physiologist 39:A53.
Zambraski, E.J., & M.J. Dunn (1993). Effects of non-steroidal anti-inflammatory drugs on renal function. In: J. H. Stewart (Eds.), Analgesic and NSAID-induced kidney disease. Oxford: Oxford University Press, pp. 147-159.
Zambraski, E.J., T.A. Rofrano, & C.D. Ciccone (1982). Effects of aspirin treatment on kidney function in exercising man. Med. Sci. Sports Exer. 14:419-423.
Zipser, R.D., J.C. Hoefs., P.F. Speckart., P.K. Zia, & R. Horton (1979). Prostaglandins: Modulators of renal function and pressor resistance in chronic liver disease. J. Clin. Endocrinol. Med. 48:895-900.